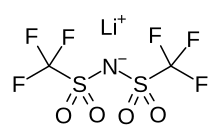
Lithium bis(trifluoromethanesulfonyl)imide
 | |
| Names | |
|---|---|
IUPAC name Lithium bis(trifluoromethylsulfonyl)azanide | |
| Other names LiTFSI | |
| Identifiers | |
CAS Number |
|
3D model (JSmol) |
|
PubChem CID |
|
RTECS number |
XR2775000 |
UNII |
|
InChI
| |
SMILES
| |
| Properties | |
Chemical formula |
LiC 2F 6NO 4S 2 |
Molar mass |
287.075 g/mol |
| Appearance |
White solid |
Odor |
odorless |
Density |
1.33 g/cm3 |
Melting point |
236 °C (457 °F; 509 K) |
Solubility in water |
soluble in water |
| Hazards | |
Safety data sheet |
[1] |
| Related compounds | |
Other anions |
Bistriflimide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
‹See TfM› | |
Infobox references | |
Lithium bis(trifluoromethanesulfonyl)imide, often simply referred to as LiTFSI, is a hydrophilic salt with the chemical formula LiC2F6NO4S2.[1] It is commonly used as Li-ion source in electrolytes for Li-ion batteries as a safer alternative to commonly used lithium hexafluorophosphate.[2] It is made up of one Li cation and a bistriflimide anion.
Because of its very high solubility in water (> 21 m), LiTFSI has been used as lithium salt in water-in-salt electrolytes for aqueous lithium-ion batteries.[3][4][5]
References
^ Pubchem. "90076-65-6 | C2F6LiNO4S2 - PubChem". pubchem.ncbi.nlm.nih.gov. Retrieved 2017-03-11..mw-parser-output cite.citation{font-style:inherit}.mw-parser-output q{quotes:"""""""'""'"}.mw-parser-output code.cs1-code{color:inherit;background:inherit;border:inherit;padding:inherit}.mw-parser-output .cs1-lock-free a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/6/65/Lock-green.svg/9px-Lock-green.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-limited a,.mw-parser-output .cs1-lock-registration a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/d/d6/Lock-gray-alt-2.svg/9px-Lock-gray-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-lock-subscription a{background:url("//upload.wikimedia.org/wikipedia/commons/thumb/a/aa/Lock-red-alt-2.svg/9px-Lock-red-alt-2.svg.png")no-repeat;background-position:right .1em center}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration{color:#555}.mw-parser-output .cs1-subscription span,.mw-parser-output .cs1-registration span{border-bottom:1px dotted;cursor:help}.mw-parser-output .cs1-hidden-error{display:none;font-size:100%}.mw-parser-output .cs1-visible-error{font-size:100%}.mw-parser-output .cs1-subscription,.mw-parser-output .cs1-registration,.mw-parser-output .cs1-format{font-size:95%}.mw-parser-output .cs1-kern-left,.mw-parser-output .cs1-kern-wl-left{padding-left:0.2em}.mw-parser-output .cs1-kern-right,.mw-parser-output .cs1-kern-wl-right{padding-right:0.2em}
^ Kalhoff, Julian; Bresser, Dominic; Bolloli, Marco; Alloin, Fannie; Sanchez, Jean-Yves; Passerini, Stefano (2014-10-01). "Enabling LiTFSI-based Electrolytes for Safer Lithium-Ion Batteries by Using Linear Fluorinated Carbonates as (Co)Solvent". ChemSusChem. 7 (10): 2939–2946. doi:10.1002/cssc.201402502. ISSN 1864-564X.
^ Suo, Liumin; Borodin, Oleg; Gao, Tao; Olguin, Marco; Ho, Janet; Fan, Xiulin; Luo, Chao; Wang, Chunsheng; Xu, Kang (2015-11-20). ""Water-in-salt" electrolyte enables high-voltage aqueous lithium-ion chemistries". Science. 350 (6263): 938–943. doi:10.1126/science.aab1595. ISSN 0036-8075. PMID 26586759.
^ Smith, Leland; Dunn, Bruce (2015-11-20). "Opening the window for aqueous electrolytes". Science. 350 (6263): 918–918. doi:10.1126/science.aad5575. ISSN 0036-8075. PMID 26586752.
^ Suo, Liumin; Borodin, Oleg; Gao, Tao; Olguin, Marco; Ho, Janet; Fan, Xiulin; Luo, Chao; Wang, Chunsheng; Xu, Kang (2015-11-20). ""Water-in-salt" electrolyte enables high-voltage aqueous lithium-ion chemistries". Science. 350 (6263): 938–943. doi:10.1126/science.aab1595. ISSN 0036-8075. PMID 26586759.
This article about an organic compound is a stub. You can help Wikipedia by expanding it. |

Comments
Post a Comment